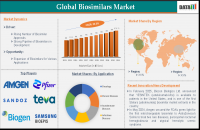
(EMAILWIRE.COM, April 09, 2025 ) Global Biosimilars market size reached US$ 22.58 billion in 2024 and is expected to reach US$ 171.79 billion by 2033, growing at a CAGR of 25.5% during the forecast period 2025-2033.
Download Free Sample: https://www.datamintelligence.com/download-sample/biosimilars-market
Market Dynamics
Surge in Biosimilar Approvals Accelerates Market Expansion
The growing number of biosimilar approvals is playing a pivotal role in driving the global biosimilars market forward. As regulatory agencies continue to greenlight biosimilars across a broader range of biologics, the market is expanding into diverse therapeutic areasincluding oncology, autoimmune disorders, chronic diseases, diabetes, and hematology.
A recent example highlighting this trend came in March 2025, when Celltrion announced that the U.S. Food and Drug Administration (FDA) approved OMLYCLO (omalizumab-igec). It is the first and only biosimilar designated as interchangeable with XOLAIR (omalizumab), indicated for the treatment of moderate to severe persistent asthma, chronic rhinosinusitis with nasal polyps (CRSwNP), IgE-mediated food allergy, and chronic spontaneous urticaria (CSU).
Market Segments
By Product Type (Monoclonal Antibodies, Recombinant Human Growth Hormone (rhGH), Insulin, Anti-coagulants, Erythropoietin, Granulocyte Colony Stimulating Factor, Follitropin, Interferons, Others)
By Application (Oncology, Chronic Diseases, Autoimmune Diseases, Infectious Diseases, Growth Hormone Deficiency, Hematology, Others)
By Region (North America, Latin America, Europe, Asia Pacific, Middle East, and Africa)
Read Our Report: https://www.datamintelligence.com/research-report/biosimilars-market
Market Regional Share
North America Poised to Lead the Global Biosimilars Market
North America is expected to dominate the global biosimilars market, driven by strong regulatory support, a growing number of product approvals, and the presence of major pharmaceutical players.
The U.S. Food and Drug Administration (FDA) has been instrumental in shaping the regions biosimilars landscape. The introduction of the Biologics Price Competition and Innovation Act (BPCI Act) of 2009 established a clear regulatory pathway for biosimilar approvals. This framework allows biosimilars to be approved based on their similarity to reference biologics, eliminating the need to duplicate extensive clinical trials.
A landmark moment came in 2015 with the FDA’s approval of Zarxio (a biosimilar to Neupogen), marking the entry of biosimilars into the U.S. market. Since then, 71 biosimilars have received FDA approval, with the most recent being Omlyclo (omalizumab-igec), approved on March 7, 2025.
North America also benefits from being home to pharmaceutical giants such as Amgen, Pfizer, Sandoz, and Boehringer Ingelheimall heavily invested in biosimilar development. Products like Amjevita (adalimumab biosimilar by Amgen) and Inflectra (infliximab biosimilar by Pfizer) are already making a significant impact in the U.S. market.
Additionally, in March 2025, Celltrion announced the U.S. launch of STEQEYMA (ustekinumab-stba), a biosimilar to STELARA, following its FDA approval in December 2024. STEQEYMA is approved for all the same indications as its reference product, offering consistent treatment options for both patients and providers.
With a robust regulatory framework, accelerating approvals, and a strong industry presence, North America is set to maintain a leading position in the global biosimilars market.
Key Market Players
Key players are Amgen Inc., Pfizer Inc., Sandoz Group AG, Teva Pharmaceuticals USA, Inc., Biogen, Biocon Biologics Inc., Boehringer Ingelheim International GmbH, Samsung Bioepis, Dr. Reddys Laboratories Ltd and Fresenius Kabi AG.